Translating Science
Leveraging our unparalleled expertise in diseases of the immune system and CXCR4 biology, we aim to bring innovative treatments to people with chronic neutropenic disorders and WHIM syndrome.
What is Chronic Neutropenia?
Chronic neutropenia is defined as periods lasting more than three months, persistently or intermittently, where there are low levels of certain white blood cells called neutrophils, which play an important role in the body’s defense against pathogens. Chronic neutropenic disorders are rare blood conditions where people of all ages experience low levels of neutrophils and tend to be at greater risk of developing infections and certain types of cancer. Depending on the levels of circulating neutrophils in the blood, neutropenia can be categorized as mild, moderate, or severe.
Why Focus on CXCR4?
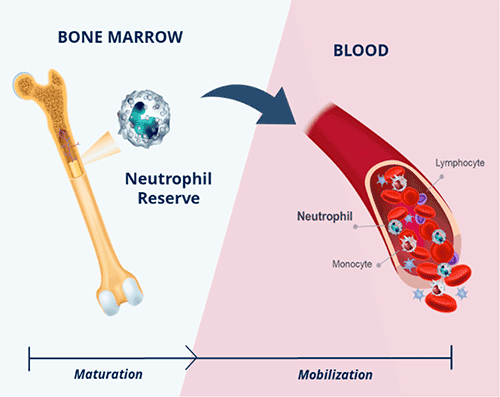
CXCR4, or C-X-C receptor type 4, is a cell receptor that helps regulate the movement of immune cells within the body. CXCR4 receptor stimulation by its ligand, CXCL12, has been shown to play a key role in the maturation and mobilization of white blood cells, including neutrophils, lymphocytes, and monocytes, into the bloodstream. Because antagonism of the CXCR4 receptor has been shown to increase the trafficking of white blood cells, we believe that therapeutic inhibition of the CXCR4/CXCL12 axis holds the potential to benefit people with a wide variety of diseases.
Our deep understanding of the biology of the CXCR4 pathway has enabled us to advance our lead candidate, mavorixafor, an orally available antagonist of the CXCR4 receptor, into clinical development. In clinical trials to date, mavorixafor has demonstrated the ability to improve the mobilization of neutrophils, lymphocytes, and monocytes into the blood stream in people with moderate to severe chronic neutropenic disorders, WHIM syndrome, and certain cancers.
We believe that successfully developing mavorixafor and providing a new therapeutic option to people with chronic neutropenic disorders has the potential to revolutionize the treatment landscape, which is currently only served by injectable therapies that have been associated with high-burden side effects.
Unparalleled Expertise in CXCR4 and Immune System Biology
In addition to our headquarters in Boston, Massachusetts, we have established a research center of excellence in Vienna, Austria. Here, our scientists not only focus on new drug discovery and on better understanding immune system dysfunction and the role of the CXCR4/CXCL12 axis in maintaining healthy immunity, but also on further studying the genetic causes of certain chronic neutropenic disorders and the mechanism of mavorixafor.
